Glioblastoma Research: Why a 2015 Drug Combination Study Still Draws Attention
A glioblastoma diagnosis is deeply difficult for both patients and families. This fast-growing form of brain cancer often continues to advance even when treated with surgery, radiation, and chemotherapy. Because of that reality, researchers around the world keep searching for better strategies, including new uses for drugs that are already approved for other conditions.
One laboratory finding from several years ago still stands out. It suggested that two familiar types of medication might affect glioblastoma cells in an unexpected way. Although the research was early and far from routine clinical use, it remains an interesting example of how everyday medicines can reveal surprising possibilities in cancer science.
Glioblastoma at a Glance
Glioblastoma is among the most aggressive primary brain tumors seen in adults. It tends to grow quickly and can disrupt vital brain functions, which often makes daily life more difficult for the person affected.
Current treatment usually involves:
- Surgery
- Radiation therapy
- Chemotherapy
Even with these standard approaches, long-term outcomes are still limited, which is why glioblastoma remains a major focus of medical research. One reason it is especially hard to treat is that its cells are highly adaptable. They can survive stress and often spread into nearby healthy brain tissue, making complete removal very challenging.
For many families, the experience brings both hope and uncertainty. That is why even early laboratory discoveries can feel important, especially when they offer a fresh angle on how tumor cells might be targeted.
Autophagy: The Cell’s Recycling System
Autophagy is a natural process cells use to clear out damaged material and recycle internal components. You can think of it as a built-in cleanup and repair mechanism. Under normal conditions, this process helps cells stay functional during stress or when nutrients are limited.
But autophagy is not always straightforward. In some situations, it protects cells. In others, if activated too strongly, it may cause cells to break down too much of their own structure.
That dual role is one reason scientists have studied autophagy so closely in cancer. If tumor cells rely on this system to survive stress, then changing the process might influence whether they continue living or begin to self-destruct.
This idea became especially interesting when researchers examined whether two well-known drug classes could push autophagy beyond a safe limit inside glioblastoma cells.
The 2015 Mouse Study That Sparked Interest
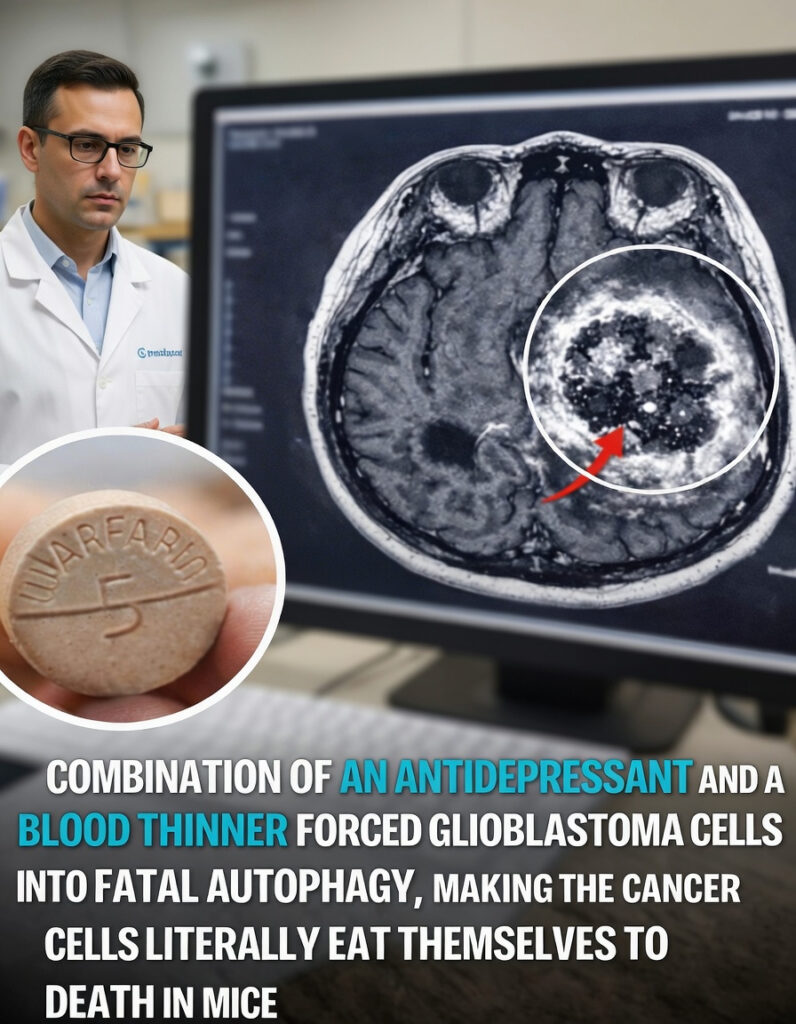
In 2015, scientists at the Swiss Federal Institute of Technology in Lausanne (EPFL) published a study in Cancer Cell that drew wide attention. They tested a tricyclic antidepressant called imipramine together with ticlopidine, an antiplatelet drug often described broadly as a blood thinner, in mouse models of early-stage human glioblastoma.
A key clarification is important here: some online illustrations use more familiar anticoagulants such as warfarin, but the actual study focused on ticlopidine.
The researchers found that the drug pair appeared to hyper-activate autophagy within glioblastoma cells. Instead of simply helping the cells cope with stress, the process became so intense that the tumor cells began breaking down their own internal structures.
Used separately, each drug produced only a limited effect in the models. Used together, however, they showed a synergistic action. In the mice, survival time was nearly doubled compared with untreated groups. Even so, the tumors were not eradicated. The treatment slowed progression rather than producing a cure.
Simple Breakdown of the Findings
| Treatment | Effect in Mouse Models | Survival Impact |
|---|---|---|
| Antidepressant alone | Mild increase in autophagy | No meaningful improvement |
| Ticlopidine alone | Mild increase in autophagy | No meaningful improvement |
| Combination of both drugs | Strong hyper-activation of autophagy | Survival nearly doubled |
This comparison helps explain why the results generated interest. The medications appeared to influence different points in the same cellular pathway, removing some of the usual limits on autophagy and pushing tumor cells past a tolerable threshold.
How the Two Drugs Appeared to Work Together
The science is complex, but the basic idea is fairly simple. The antidepressant increased signaling in one part of the autophagy pathway, while ticlopidine affected another part. Together, they created an overload effect.
Instead of helping the cancer cells adapt, this stronger activation seemed to push them into self-destruction. In these early experiments, normal cells appeared to be far less affected, which added to the excitement surrounding the findings.
This is also why drug repurposing attracts so much interest. When researchers test older, established medications for new diseases, they can build on safety information that already exists. These drugs are often easier to study, more affordable, and more familiar than completely new compounds.
Still, the researchers were careful in their interpretation. They did not present the approach as a cure. Their conclusion was that it delayed disease progression and might eventually need to be paired with other anti-cancer treatments to have broader value.
Why Drug Repurposing Matters in Glioblastoma Research
Using approved drugs in new ways is one of the more practical strategies in medical science. It can shorten part of the research process because those medications have already been studied for safety in other settings.
For a cancer as difficult as glioblastoma, that makes repurposing especially appealing. It offers a route to explore potential treatment ideas without starting from zero.
Studies like this often create a mix of optimism and caution. That response is reasonable. Early findings can be intriguing, but careful testing is essential before anything can be considered useful for real-world patient care.
Practical Ways to Stay Informed and Support Overall Health
This research remains experimental and should not be treated as a recommended therapy. Still, there are constructive steps people can take to stay informed and support general well-being while science continues to move forward.
A 5-Step Plan You Can Start Now
-
Speak openly with your medical team
- Bring articles or research findings to appointments.
- Ask how they relate, if at all, to current evidence-based treatment options.
-
Use trusted information sources
- Follow reputable organizations such as the National Cancer Institute.
- Read peer-reviewed medical publications when possible.
- Be cautious with claims shared on social media.
-
Stay physically active
- Aim for about 30 minutes of light movement, such as walking, on most days if your doctor approves.
- Regular activity supports overall health and recovery.
-
Choose nutrient-dense foods
- Focus on colorful vegetables, healthy fats, and lean protein sources.
- Good nutrition supports the body during stressful periods.
-
Protect your sleep
- Try to get 7 to 9 hours of rest each night.
- Consistent sleep helps natural repair and recovery systems function more effectively.
These steps are not substitutes for treatment, but they can help people feel more informed and engaged during a difficult time.
Research Limitations and What Still Needs to Be Proven
It is important to keep the findings in perspective. This was a mouse study, and it used early-stage glioblastoma models. Results seen in animals do not always translate to humans.
So far, no large clinical trials have confirmed that the same drug combination produces similar benefits in people with glioblastoma. Human tumors are more complex, and the body’s response to treatment can differ significantly from what happens in laboratory models.
The original researchers also noted that this approach would likely need to be combined with additional anti-cancer therapies for greater effect. That point helps keep the discussion realistic and grounded in evidence rather than hype.
Conclusion
The 2015 study opened an intriguing line of research into how an antidepressant and an antiplatelet drug might alter glioblastoma cells through excessive autophagy. It did not provide a cure, but it did show that common medications can sometimes interact with cancer biology in unexpected ways.
That is part of what makes cancer research so compelling. Sometimes progress comes not only from brand-new drugs, but also from re-examining familiar ones with fresh scientific questions. Until stronger evidence emerges, the most practical approach remains staying informed, following standard medical guidance, and supporting overall health as research continues.
Frequently Asked Questions
What happens during autophagy in this kind of research?
Autophagy is the cell’s internal recycling system. In this study, researchers tried to push that process so strongly in glioblastoma cells that it became destructive rather than protective. The idea was that over-activating autophagy could cause tumor cells to break down their own components.
Did the drug combination cure glioblastoma in the study?
No. The combination did not eliminate the tumors in the mouse models. It slowed tumor progression and extended survival time, but it was not a cure.
Why was the study considered important?
It suggested that two existing drug classes might work together in a synergistic way against glioblastoma cells. That finding supported further interest in drug repurposing and autophagy-based cancer research.
Can patients use this drug combination now?
This research should not be viewed as a current treatment recommendation. The findings came from animal models, and similar benefits have not been proven in large human clinical trials.
Why do researchers keep studying older drugs for new diseases?
Older medications already have known safety histories, which can make research faster and more practical. For difficult cancers like glioblastoma, repurposed drugs may offer useful leads that deserve further investigation.