Ramipril Recall: What Patients Need to Know and What to Do Next
If you or someone close to you takes ramipril capsules every day to manage blood pressure or support heart health, news of a nationwide recall can be alarming. It is completely understandable to feel concerned when you start wondering whether the medication in your home is affected and whether immediate action is necessary. For many people, staying consistent with daily treatment is essential, so any disruption can feel stressful.
The good news is that the recall announced by Lupin Pharmaceuticals in coordination with the FDA is a precautionary measure. Most ramipril products currently in use are not involved.
Still, the details matter. Knowing exactly what happened and what steps to take can help you respond calmly and safely.
What Happened in the Ramipril Recall?
The U.S. Food and Drug Administration, together with Lupin Pharmaceuticals, has issued a voluntary nationwide recall covering more than 600,000 bottles of ramipril capsules. The concern is linked to the active ingredient, which may have been sourced from a manufacturing site in India that was neither approved nor inspected.
Regulators have described this recall as preventive. At this time, there have been no reports of illness or harmful side effects connected to the recalled lots.
This situation highlights two important realities:
- The pharmaceutical supply chain is highly complex and often global
- Safety systems are designed to catch potential issues before they become larger problems

Which Ramipril Capsules Are Included?
Not all ramipril capsules are affected. The recall applies only to selected lots distributed by Lupin Pharmaceuticals.
Check your bottle for the following details:
- Ramipril capsule strengths: 2.5 mg, 5 mg, and 10 mg
- Bottle sizes: 90-count, 100-count, and 500-count
- Expiration dates: through July 2026
Many ramipril prescriptions filled at pharmacies come from different manufacturers or from Lupin lots that are not part of the recall. That means most supplies remain available and safe to use.
Why This Recall Matters to Daily Users
Medication recalls can be unsettling, especially when the medicine is part of your everyday routine. Even the possibility of a quality concern can create extra pressure when you are already focused on protecting your health.
However, one point is especially important: do not stop taking ramipril suddenly unless a healthcare professional tells you to do so. Abruptly stopping blood pressure medication may cause your blood pressure to rise again, which could create greater health risks.
That is why guidance from your doctor, pharmacist, and the FDA is so important during a recall like this.
Do Not Stop Ramipril Without Medical Guidance
Stopping ramipril on your own is not advised. Your body may rely on the medication’s ongoing effect to help maintain stable blood pressure and support heart function.
If your bottle is affected, your pharmacy or healthcare provider can usually help you transition smoothly. In many cases, a replacement can be arranged quickly and without additional cost.
The best approach is simple:
- Stay calm
- Confirm whether your medication is affected
- Contact a professional before making any changes
What to Do Right Now If You Take Ramipril
Reviewing your medication bottle only takes a few minutes and can give you peace of mind. Use the following steps:
-
Check the label carefully
- Look for the manufacturer name: Lupin Pharmaceuticals
- Confirm the strength, bottle size, lot number, and expiration date
-
Compare your bottle with the official recall information
- Visit the FDA recall page
- Check Lupin Pharmaceuticals’ official website for the affected lot numbers
-
Contact your pharmacy or healthcare provider if your bottle matches
- Let them know your medication appears to be part of the recall
- Ask what replacement options are available
-
Request a replacement supply
- In most cases, pharmacies can provide a substitute without interrupting your treatment
- Many replacements are handled at no extra cost
-
Speak with your doctor if needed
- Your provider may recommend continuing with a new ramipril supply
- In some cases, another medication in the same class may be considered
This process helps you stay on top of the situation without putting your health routine at risk.
What to Expect When You Contact the Pharmacy
Pharmacists are familiar with medication recalls and usually have a clear process in place. If your ramipril is part of the recall, the pharmacy can often replace it promptly so you do not miss doses.
Your doctor may also decide that another treatment option is appropriate, depending on your health history and current needs. Because every patient is different, personalized advice is always the safest route.
Daily Habits That Support Your Health During a Recall
While you sort out recall details, a few simple habits can help you stay organized and confident with your medication routine:
- Keep ramipril in its original container
- Store it in a cool, dry place away from direct sunlight
- Monitor your supply so you do not run out unexpectedly
- Save lot numbers and prescription details for quick reference
- Check in with your healthcare provider regularly
- Use a dependable home blood pressure monitor between appointments
- Support your treatment plan with exercise, balanced meals, and consistent sleep
These practical steps can make medication management easier, especially when unexpected news arises.
Why This Recall Reflects a Larger Safety System
This recall is also a reminder of how much work goes into maintaining medication quality. Drug manufacturers and regulatory agencies continuously monitor the supply chain to identify issues early and respond quickly.
Preventive recalls like this are part of that safety process. Rather than waiting for harm to occur, regulators act when there is reason for concern. That kind of response helps protect patients and maintain trust in the system.
For most people taking ramipril, the outcome will likely be reassuring: their medication will not be part of the recalled lots.
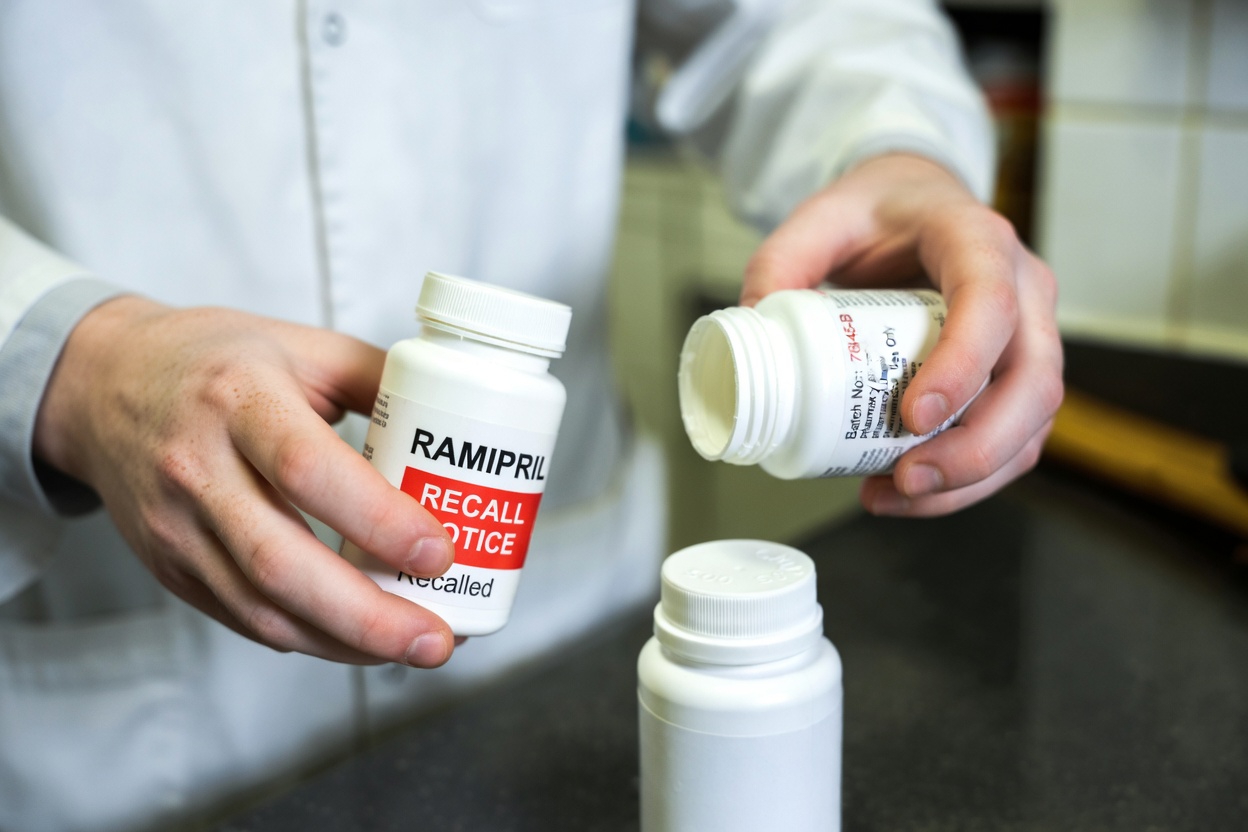
Final Thoughts: Stay Informed and Stay in Control
Hearing about a ramipril recall can feel overwhelming at first, but the situation is manageable when you know what to do. Start by checking your bottle, confirm the lot information through official sources, and contact your pharmacist or doctor if you have any concerns.
Your health remains the priority. With support from the FDA, your pharmacy, and your healthcare provider, you can handle this recall safely and keep your treatment plan on track.
Frequently Asked Questions
1. Should I throw away my ramipril immediately if I think it may be recalled?
No. First verify the lot details and speak with your pharmacist or doctor before discarding the medication or stopping treatment. They can tell you the safest next step.
2. Where can I find the most current list of recalled lot numbers?
The most reliable sources are:
- The official FDA recall page
- The official Lupin Pharmaceuticals website
These sources provide the latest updates available.
3. Will I have to pay for a replacement if my bottle is recalled?
In many situations, pharmacies can arrange a replacement at no additional cost. Contact your pharmacy directly to confirm how it will be handled in your specific case.
Disclaimer
This article is provided for informational purposes only and does not replace professional medical advice. Always speak with your doctor, pharmacist, or other qualified healthcare provider before making changes to your medication routine. Information in this article is based on FDA and manufacturer announcements available as of March 2026.



